I often see TV commercials about Xarelto®, one of the new anticoagulants, late at night when I’m trying to wind down after a full day. (I am a “Forensic Files”-ophile.) The people in the Xarelto® ads say they have “Afib” and speak ominously about a “higher risk of stroke” because of this heart-rhythm disorder. I sometimes find myself wondering how many viewers, upon hearing that, understand why that is. Do you?
Xarelto® (rivaroxaban) and other so-called latest-generation anticoagulants, such as Eliquis® and Pradaxa®, are still on patent and will not be available in generic form for years yet. These anticoagulants—incorrectly described as “blood thinners”—are typically touted as a superior alternative to warfarin (Coumadin®), which has been in use for more than 60 years, because patients taking them do not have to have their blood tested regularly nor do they have to restrict their diets by limiting foods high in Vitamin K, such as spinach. Vitamin K is essential to the body chemistry that results in clotting.
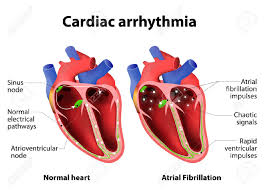
I have previously given what I call a cardiac primer about how the electricity-powered heart works. Most recently, I explained how a heart beat starts with an electrical signal emanating from the sinoatrial (SA) node in the rear of the right atrium and how impulses travel through the heart’s four chambers, enabling the heart to relax and contract, in my 2/14/18 blog about hypertension. I also covered this process in a 2/15/17 blog post. When the heart is beating as it should, it is said to be in normal sinus rhythm.
In my book, “Our Parents in Crisis: Confronting Medical Errors, Ageist Doctors, and Other Healthcare Failings,” I quoted two cardiology specialists at Johns Hopkins as saying that millions of heart muscle cells have the ability to “create their own electrical signals, [thus] disrupting the normal sinus rhythm.”
If muscle cells “misfire,” Hugh G. Calkins, M.D., and Ronald Berger, M.D., Ph.D., explain, the heart may race from a resting rate of 60 to 90 beats per minute up to 200 beats per minute, and then slow down after just a few moments. “This irregularity,” they say, “may occur hundreds of times a day, or only in several short episodes a year.”
Electrical misfirings can result in premature or so-called ectopic heartbeats, meaning that a source other than the SA node is initiating them. If a “run” of such premature beats occurs, say Calkins and Berger, the atria may start to fibrillate, or to quiver, rather than to contract forcefully. Atrial fibrillation, which I’ll abbreviate as AF, leads to an irregular and often rapid beating of the ventricles. If the ventricles don’t properly pump the blood out to the lungs and the body, blood may pool in the heart, thus promoting clot formation. A clot can migrate from the heart to the brain and cause an ischemic stroke.
Ischemia describes a condition in which blood, and therefore, oxygen, is not getting to a part of the body. It’s a reduction in essential blood supply—a deficiency—caused by an obstruction of some kind.
My Father’s AF Experience
Over a period of 10 days in 2005, my father had five episodes of AF, each lasting from four-and-a-half to 12 hours. During this period, he saw his cardiologist, who documented his AF with an ECG reading and put him on metoprolol (Toprol®), a beta blocker that slows the heart’s ventricular rate.
AF medications either control the heart’s rate or correct its rhythm. Heart rate-control medications, such as metoprolol, allow the AF to continue.
For my father, a beta blocker was a bad idea: His heart rate fell to just 40 beats per minute!
His cardiologist then switched him to flecainide (Tambocor®), an antiarrhythmic drug that stops the fibrillation and restores the heart’s sinus rhythm.
Dad had what is called paroxysmal atrial fibrillation, which Johns Hopkins Drs. Calkins and Berger describe as “a recurrent condition where the rapid heart rate and abnormal electrical signals spontaneously begin, typically last for a day or two, sometimes as long as a week, and then suddenly disappear as mysteriously as [they] began.” Paroxysmal symptoms, they say, can range from “barely noticeable to severe.”
Other types of atrial fibrillation are persistent AF, which lasts longer than a week, or is stopped short of that by a drug- or electrical-shock-induced restoration of normal rhythm, called a cardioversion; and longstanding persistent AF, which is continuous atrial fibrillation that lasts longer than a year. I have seen a fourth category described as permanent AF, in which a medical decision has been made not to restore sinus rhythm.
(I leave a full discussion of available treatment for AF for another day.)
Roughly 83 percent of all strokes are ischemic, often brought on by cerebral atherosclerosis; and 17 percent are hemorrhagic, i.e., caused by bleeding, such as when an aneurysm ruptures. AF causes about 20 percent of all ischemic strokes.
Classic stroke symptoms include slurred speech, paralysis or numbness on one side of the face or body, blurred vision, mental confusion, a severe out-of-the-blue headache, dizziness, and a loss of balance. Within four minutes of being deprived of oxygen and nutrients, brain cells begin to die.
The effects of a stroke vary from person to person and depend on its type, severity, and location. A stroke may impair a person’s movement and sensation, speech and language, vision, cognition (reasoning, memory), perception, emotional control, bowel and bladder control, and other vital body functions.
CHADS (Stroke-Risk) Scoring
Both my father’s regular cardiologist and other specialists he consulted discussed antithrombotic (anti-clot) therapy with him. If you have AF and are trying to decide whether to take an anticoagulant to thwart blood clotting in your heart, you should assess your risk of having an ischemic stroke.
Taking an anticoagulant is an everyday commitment, as well as an inconvenience and a risk. All drugs cause potential adverse effects, some of which depend on who’s taking them. Because anticoagulants interfere with blood clotting, all people taking them have to be alert to excessive bleeding.
Not only does a person on an anticoagulant bleed more upon being injured–for example, from a cut or abrasion, or from a traumatic brain injury–he or she may experience internal bleeding unknown to him. I have heard anecdotally from physicians that internal bleeding is associated with the latest-generation anticoagulants.
An electrophysiologist told Dad that because of his hypertension, which he controlled with medication; his advanced age (80); and his dilated left atrium, he was at high risk for a clot forming in his heart and should be taking the anticoagulant warfarin (Coumadin). This advice squares with the stroke-risk indexes I uncovered in my research.
While I don’t necessarily subscribe to medicine-by-the-numbers or by scoring, I do find some risk-factor indexes helpful for laypeople.
One of the most popular and simple stroke-risk indexes to use is known by the acronym, CHADS2, which abbreviates the following risk factors:
C: Congestive heart failure
H: Hypertension (above 140/90 mmHg.)
A: Age (75 or older)
D: Diabetes mellitus
S2: Stroke (prior stroke or TIA)
To compute a CHADS2 score, you assign one point to the first four risk factors and two points to the fifth, S2, which stands for prior stroke or transient ischemic attack. According to Calkins and Berger, the Johns Hopkins cardiologist-electrophysiologists whom I earlier quoted, a score of zero or one suggests a low risk of a stroke; two to three points connote a moderate risk, and any score of four or higher is a high risk. (Other physicians may evaluate a score of one as a moderate risk and any score of two or higher, a high risk.)
Physicians do not recommend anticoagulation for a low-risk AF patient, except a daily baby aspirin, if the patient chooses. The standard medical recommendation for the moderate-risk patient is an oral anticoagulant or aspirin (which also promotes bleeding); and for the high-risk patient, an oral anticoagulant, typically either warfarin or dabigatran (Pradaxa).
My father’s CHADS2 score was two. An AF patient who has a score of two and does not choose to anticoagulate purportedly has an annual stroke risk of 4.0 percent, which means that, of 100 AF patients who don’t anticoagulate, four will have a stroke.
A more recently derived index, CHA2 DS2-VASc, refines and extends CHADS2 and reportedly has proved to be a better predictor of risk. CHA2 DS2-VASc stands for:
C: Congestive heart failure or left ventricular systolic dysfunction (1 pt.)
H: Hypertension, either blood pressure consistently above 140/90 mmHg or treated hypertension on medication (1 pt.)
A2: Age 75 years or older (2 pts.)
D: Diabetes mellitus (1 pt.)
S2 : Prior stroke or TIA or thromboembolism (2 pts.)
V: Vascular disease (1 pt.)
A: 65 to 74 years of age (1 pt.)
Sc: Sex category (female, 1 pt.)
Dad’s CHA2 DS2-VASc score was three, which corresponds to a high risk and an annual risk of stroke without anticoagulation of 4.7 percent. Just as with CHADS2, any CHA2 DS2-VASc score of two or more indicates the need for oral anticoagulation.
Ann, 4/10/18